About paper-based microfluidics
Paper-based microfluidics was first introduced by Martinez and Al. [1] from the Whitesides group in 2007. The idea was to design microfluidic devices with a simpler and less expensive method, the resulting device being portable, easy to use and to dispose. The aim of this new approach was to make microfluidics affordable and usable without any laboratory for developing countries or to create an alternative to more expensive methods currently used in laboratories. The many advantages of paper-based microfluidics are detailed in this article.
The fluid flow in paper is driven by capillary forces and controlled by the porosity and geometry of the device. The porosity is defined by the choice of paper and determines the speed of the flow while the geometry is defined by the fabrication process. Paper being an hydrophilic substrate, hydrophobic barriers have to be designed to define channels and thereby flow direction. Several methods of fabrication are used to either change the shape of the substrate or its hydrophilic properties and some of these methods will be considered in the next articles.
There is several advantages of working with microfluidics paper:
– cheap and simple: it varies from one process to the next but fabrication processes are usually cheaper, simpler and requiring fewer steps when paper is used as the substrate.
– portable and robust: one of the first reasons paper-based chips were designed was to be easily carried and used outside of a laboratory [1] to enable new applications including diagnostics in the field for developing countries.
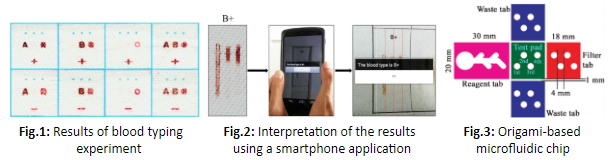
– easy to use: the devices are usually straightforward, in one piece and most of the devices use colorimetric detection [2] which is easy to read and requires no equipment. Li and Al. [3] have designed a chip that determines the blood type and shows it in writing for a clear and easy interpretation of the result: the letters A,B or O as well as a plus or a minus sign appear according to the blood type (Figure 1). Some devices are designed so that the interpretation can be done by a smartphone application [4] [5] and directly sent to doctors to make it easier for patients (Figure 2).
– disposable: paper being easily flammable, it is possible to dispose of the chips by inceneriting them [6] [7]. This represents a effective method of disposal requiring no specific facility.
Paper-based microfluidics is often used for its simplicity and low cost but also for the new functions it offers. It is possible to use the properties of paper to fold it and form 3D devices [8][9] (Figure 3), use solid reagents and a pen to carry and deposit it on paper by drawing [10], use the paper for storage and immobilization of reagents without requiring specific surface treatment. Filtering can be achieved by the choice of an appropriate pores size and can be used to separate plasma from whole blood [11] for example.
It is also possible to use paper in addition to other materials such as glass [12] or PDMS [13] to take advantage of paper’s properties while maintaining a high resolution and be able to design more complex chips. The main properties of paper that are exploited in hybrid devices is the ability of paper to contain and immobilize biosensors.
To conclude, paper microfluidics is well-suited to design diagnostic tests that respect the “ASSURED” characteristics set by the World Health Organisation: Affordable, Sensitivity, Specificity, User-friendly, Rapid and robust, Equipment-free and Deliverable.
[1]: Martinez, A. W., Phillips, S. T., Butte, M. J., & Whitesides, G. M. (2007). Patterned Paper as a Platform for Inexpensive, Low-Volume, Portable Bioassays. Angewandte Chemie International Edition, 46(8), 1318–1320.
[2]: Paper microfluidic devices: a review 2017,elveflow, 2017, available at https://www.elveflow.com/microfluidic-tutorials/
microfluidic-reviews-and-tutorials/paper-microfluidic-devices-a-review-2017/
[3]: M. Li, J. Tian, M. Al-Tamimi, and W. Shen, “Paper-Based Blood Typing Device That Reports Patient’s Blood Type ‘in Writing,’” Angewandte Chemie International Edition, vol. 51, no. 22, pp. 5497–5501, Apr. 2012.
[4]: L. Guan, J. Tian, R. Cao, M. Li, Z. Cai, and W. Shen, “Barcode-Like Paper Sensor for Smartphone Diagnostics: An Application of Blood Typing,” Analytical Chemistry, vol. 86, no. 22, pp. 11362–11367, Oct. 2014.
[5]: K. Yamada, H. Shibata, K. Suzuki, and D. Citterio, “Toward practical application of paper-based microfluidics for medical diagnostics: state-of-the-art and challenges,” Lab on a Chip, vol. 17, no. 7, pp. 1206–1249, 2017.
[6]: L. Busa, S. Mohammadi, M. Maeki, A. Ishida, H. Tani, and M. Tokeshi, “Advances in Microfluidic Paper-Based Analytical Devices for Food and Water Analysis,” Micromachines, vol. 7, no. 5, p. 86, May 2016.
[7]: S. Altundemir, A. K. Uguz, and K. Ulgen, “A review on wax printed microfluidic paper-based devices for international health,” Biomicrofluidics, vol. 11, no. 4, p. 41501, Jul. 2017.
[8]: L. Ge, S. Wang, X. Song, S. Ge, and J. Yu, “3D Origami-based multifunction-integrated immunodevice: low-cost and multiplexed sandwich chemiluminescence immunoassay on microfluidic paper-based analytical device,” Lab on a Chip, vol. 12, no. 17, p. 3150, 2012.
[9]: J. Qi et al., “Three-dimensional paper-based microfluidic chip device for multiplexed fluorescence detection of Cu2+ and Hg2+ ions based on ion imprinting technology,” Sensors and Actuators B: Chemical, vol. 251, pp. 224–233, Nov. 2017.
[10]: H. T. Mitchell et al., “Reagent pencils: a new technique for solvent-free deposition of reagents onto paper-based microfluidic devices,” Lab on a Chip, vol. 15, no. 10, pp. 2213–2220, 2015.
[11]: X. Yang, O. Forouzan, T. P. Brown, and S. S. Shevkoplyas, “Integrated separation of blood plasma from whole blood for microfluidic paper-based analytical devices,” Lab Chip, vol. 12, no. 2, pp. 274–280, 2012.
[12]: P. Zuo, X. Li, D. C. Dominguez, and B.-C. Ye, “A PDMS/paper/glass hybrid microfluidic biochip integrated with aptamer-functionalized graphene oxide nano-biosensors for one-step multiplexed pathogen detection,” Lab on a Chip, vol. 13, no. 19, p. 3921, 2013.
[13]: Dou, M., Dominguez, D. C., Li, X., Sanchez, J., & Scott, G. (2014). A Versatile PDMS/Paper Hybrid Microfluidic Platform for Sensitive Infectious Disease Diagnosis. Analytical Chemistry, 86(15), 7978–7986.
[14]: C. S. Kosack, A.-L. Page, and P. R. Klatser, “A guide to aid the selection of diagnostic tests,” Bulletin of the World Health Organization, vol. 95, no. 9, pp. 639–645, Jun. 2017.
contact@blackholelab.com or phone: +33(0).782.831.207